
|
HYBRID PRIMATES |
LEMURS
Lemur taxonomy is controversial and species have been reclassified on several occasions - I have added notes mapping old and new names to each other. In "The Variation Of Animals And Plants Under Domestication" Charles Darwin noted: "Several members of the family of Lemurs have produced hybrids in the Zoological Gardens."
The White-headed Lemur (Eulemur albifrons) has hybridised with the Common Brown Lemur (E. fulvus) and both male and female hybrids appear to be at least partially fertile (there are records of a male hybrid siring offspring). The White-headed Lemur has also interbred in captivity with the Black Lemur (E. macaco), and with the Mongoose lemur (E. mongoz).
The Grey-headed Lemur (E. cinereiceps) (formerly called the White-collared Lemur - E. albocollaris) has bred with the Red-collared Brown Lemur (E. collaris). The Grey-headed Lemur has also bred with the Common Brown Lemur (E. fulvus) producing fertile female hybrids; one such hybrid produced offspring with a Black Lemur (E. macaco) male. The Grey-headed Lemur can interbreed with the Red-fronted Lemur (E. rufifrons) producing fertile male and female offspring; one such hybrid bred with the Common Brown Lemur. The Grey-headed Lemur has hybridised with the Black Lemur.

A Red-fronted Lemur (E. rufifrons , described as E. fulvus rufus) x White-Collared Brown Lemur (E. albocollaris) hybrid zone at Andringitra, Madagascar was examined using DNA studies. When the DNA markers of putative hybrids were examined, 18 of 21 were found to contain markers from both E. albocollaris and E.f. rufus populations. The remaining three individuals were found to contain only markers for E. albocollaris. The results indicate that the population at Andringitra is a hybrid population between the two species. The White Collared Brown Lemur was reclassified as the Gray-headed Lemur (Eulemur cinereiceps) in 2008. The Red-fronted Lemur is now Eulemur rufifrons.
Wyner Yael M; Johnson Steig E; Stumpf Rebecca M; Desalle Rob. "Genetic assessment of a white-collared x red-fronted lemur hybrid zone at Andringitra, Madagascar." American journal of primatology 2002;57(2):51-66.
Johnson, S. E. (2002). Ecology and Speciation in Brown Lemurs: White-collared Lemurs (Eulemur albocollaris) and Hybrids (Eulemur albocollaris X Eulemur fulvus rufus) in Southeastern Madagascar. PhD dissertation. University of Texas.
Red-collared Brown Lemur (E. collaris, formerly known as Lemur xanthomystax) has hybridised with the following: with the Common Brown Lemur (E. fulvus), with the Black Lemur (E. macaco) (sterile male hybrids, in fact Black Lemur hybrids of both sexes are normally sterile according to Horvath & Willard), with the Mongoose Lemur (E. mongoz, formerly known as Lemur nigrifrons – Black-headed Lemur), and with the Red-fronted Lemur (E. rufifrons) (some hybrids fertile and able to back-cross to a parent species).
Julie E. Horvath & Huntington F. Willard (2007) Primate comparative genomics: lemur biology and evolution, Vol 23, Issue 4, P173-182, April 01, 2007
The Crowned Lemur (E. coronatus) has bred with the Black Lemur (E. macaco) where the species come into contact. The Black Lemur (E. macaco) interbreeds freely with the Blue-eyed Black Lemur (Sclater’s Lemur) (E. flavifrons) and the offspring always have orange eyes. Until 2008, the Blue-eyed Black Lemur was considered a subspecies (E. macaco flavifrons) of the Black Lemur.
S. Warter, Y. Rumpler "Cytogenetic study of a female Lemur coronatus × Lemur macaco hybrid" Faculté de Médecine, Institut d'Embryologie, 67085 Strasbourg Cédex, France
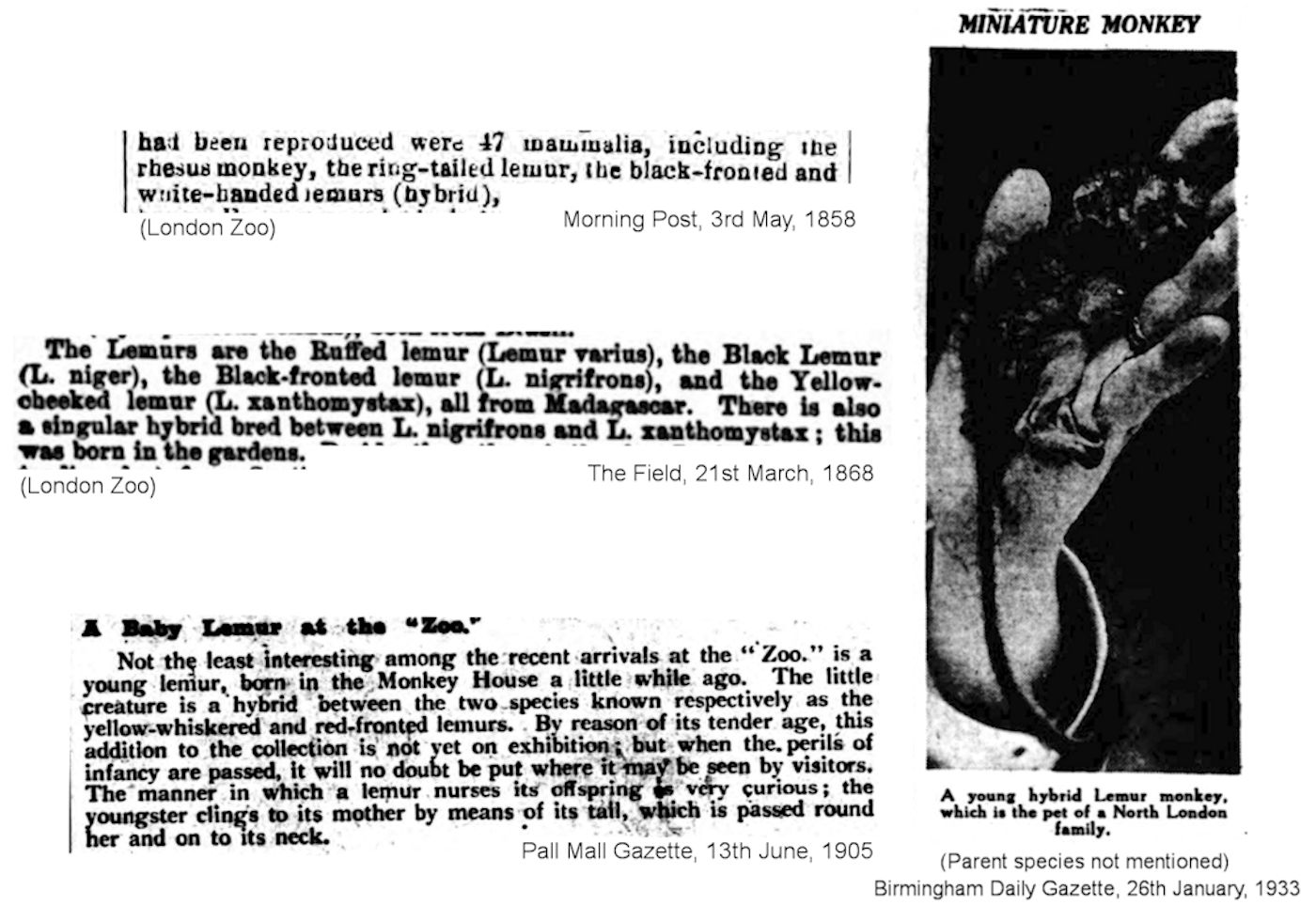
The Common Brown Lemur (E. fulvus) and the Back Lemur (E. macaco) frequently hybridised in captivity. The Common Brown Lemur has also bred with the Mongoose Lemur (E. mongoz), with the Red-bellied Lemur (E. rubriventer) (Asson Zoo, male hybrids were sterile), with the Red-fronted Lemur (E. rufifrons), and with Sanford's Brown Lemur (E. sanfordi).
Jekielek, J. (2004). Hybridization of Brown Lemurs at Berenty Reserve, Madagascar. Masters Abstracts (42(2)), p.526.
Rumpler Y. Dutrillaux B. Chromosomal evolution in Malagasy lemurs. V. Chromosomal banding studies of Lemur fulvus albifrons, Lemur rubriventer and its hybrids with Lemur fulvus fulvus. Folia Primatol. (Basel). 1980; 33: 253-261
Tanaka, M. (2007). Habitat use and social structure of a brown lemur hybrid population in the Berenty Reserve, Madagascar. American Journal of Primatology 69, pp.1189–1194.
The Black Lemur (E. macaco) had bred with the Mongoose Lemur (E. mongoz), but the hybrid died at a few days old. London Zoo exhibited a hybrid of Black Lemur x Red-fronted Lemur (E. rufifrons) that did not resemble either parent, lacking the sire’s facial fringe and the dam’s white forehead markings, instead having a dark grey-black forehead. In some resects it resembled the Common Brown Lemur (E. fulvus) which is not surprising because the Red-fronted Lemur, Red Lemur (E. rufus) and Common Brown Lemur were formerly lumped into a single species and the mother may have been a hybrid of Common Brown Lemur with one of the Red Lemur species.
Mongoose Lemur (E. mongoz) x Red-fronted Lemur (E. rufifrons), and Mongoose Lemur x Common Brown Lemur (E. fulvus) hybrids occur in the wild. The females appear to be fertile, and able to backcross with a parental species.
D. Curtis and A. Zaramody carried out a 10-month study on mongoose lemurs at Anjamena in northwestern Madagascar (Curtis 1997). They observed animals with colouration intermediate between the Mongoose Lemur (E. mongoz) and Red-fronted Lemur (E.f. rufus). This suggested there was a hybrid zone where the two species interbred. This was substantiated by the existence of a Brown lemur that had the mitochondrial (maternal) DNA of a mongoose lemur (Zaramody & Pastorini 2001).
Curtis DJ (1997) The Mongoose Lemur (Eulemur mongoz): A Study in Behaviour and Ecology. Dissertation, Universität Zürich.
Zaramody A & Pastorini J (2001) Indications for hybridisation between red-fronted lemurs (Eulemur fulvus rufus) and mongoose lemurs (E. mongoz) in northwest Madagascar. Lemur News 6:28-31
Pastorini J, Zaramody A, Curtis DJ, Nievergelt CM & Mundy NI (2009) Genetic analysis of hybridization and introgression between wild mongoose and brown lemurs. BMC Evolutionary Biology 9:32
A Red-bellied Lemur (E. rubriventer x Red-fronted Lemur (E. rufifrons) hybrid has occurred at Woburn Safari Park. The Red Lemur/Northern Red-Fronted Lemur/Rufous Brown Lemur (E. rufus) was formerly considered a subspecies of the Common Brown Lemur (E. fulvus). Until 2001, E. rufus was considered a subspecies of the E. fulvus, and then in December 2008 E. rufus was split into two separate species: E. rufus (northwestern Madagascar) and E. rufifrons (southwest and eastern Madagascar). Some historical accounts of Red-fronted Lemur (E. rufifrons) may be hybrids of Red Lemur (E. rufus or E. fulvus).
The Eastern Lesser Bamboo Lemur/Grey Gentle Bamboo Lemur (Hapalemur griseus) has formed hybrids with several other species: with the Lac Alaotra Bamboo Lemur/Bandro (H. alaotrensis) in the wild, with the Southern Lesser Bamboo Lemur (H. meridionalis) interbreed freely in the wild, with the Western (or Sambirano) Lesser Bamboo Lemur (H. occidentalis) in the wild and with the Greater bamboo Lemur (Prolemur simus) in the wild.
The Northern Sportive Lemur (Lepilemur septentrionalis) was thought to have four subspecies that interbreed in the wild to form fertile hybrids despite different chromosome counts, but this was reduced to 2 species, L. s. septentrionalis and L. s. ankaranensis, in 2001. L. s. ankaranensis was then raised to full species status (Ankarana Sportive Lemur). The Northern Sportive Lemur (L. septentrionalis) is now considered to be two separate species complexes probably caused by chromosomal rearrangements in one of the L. septentrionalis evolutionary lineages. A species complex may represent an early stage after speciation or may be due to frequent hybridisation so that the speciation process has stalled (until some other barrier to reproduction occurs).
Red-ruffed Lemur (Varecia rubra) can produce fertile hybrids with the Black-and-white Ruffed Lemur (V. variegata). During the 19th and 20th centuries these were formerly considered subspecies - Varecia variegata rubra and V. v. variegate - although a natural hybrid zone has never been documented, but in 2001 genetic research determined that they were full species. Hybrids were common in captivity (London Zoo Jersey Zoo) and also occur in a contact zone in the wild although hybridization is the exception rather than the rule. Historically, the confluence of the Vohimara and Antainambalana Rivers may have been a zone of hybridization between these two species (Garbutt, Nick (2007). Mammals of Madagascar, A Complete Guide. A&C Black Publishers). The hybrids have black caps and bellies with variable dark markings. The pale areas vary from reddish to light brown. Hybrids were bred at London Zoo in the 1970s/80s from a female Black-and-white Ruffed Lemur x male Red Ruffed Lemur at a time when these were rare in captivity. When the female died, the fertile female offspring were backcrossed to their father and a number of F2 hybrids lacked fur, possibly due to inbreeding causing recessive genes to emerge.
Vasey, Natalia.; Tattersall, Ian. (2002) Do ruffed lemurs form a hybrid zone? : distribution and discovery of Varecia, with systematic and conservation implications. American Museum novitates ; no. 3376
The Crowned Sifaka (Propithecus coronatus) can form hybrids with Van Decken’s Sifaka (P. deckenii) Van Decken’s Sifaka can form hybrids with Verreaux’s Sifaka (P. verreauxi).
Gray, A.P.: Mammalian Hybrids. Second edition. A Check-List with Bibliography. Commonwealth Agricultural Bureaux, Farnham Royal, Slough, UK, 1972.
Djlelati R. et al. Meiotic study of hybrids in the genus Eulemur and taxonomic considerations. Am. J. Primatol. 1997; 42: 235-245
Moses M.J. et al. Synaptonemal complex analysis of heteromorphic trivalents in lemur hybrids. Chromosoma. 1979; 70: 141-160
Rumpler Y. Warier S. • Ratomponirina Ch. • Hauwy M. • Ishak B. (1985) Cytogenetic Study of Complex Hybrids in the Genus Lemur (Primates Prosimians) , Foli Primatol, 108-116.
BABOONS
Most Baboon species seem able to interbreed and the female F1 hybrids are frequently fertile. In ther wild, there are several hybrid zones between species.
The Olive/Anubis Baboon (Papio anubis) x Yellow Baboon (P. cynocephalus) hybridise naturally, forming a cline (series of intermediates) between the two species. Olive/Anubis Baboon × Hamadryas Baboon (P. hamadryas) hybrids occur, but the hybrids appear viable only between male Olive/Anubis Baboon and female Yellow Baboon. Olive/Anubis Baboon x Guinea Baboon (P. papio) are likely to hybridise in the wild. Tentative hybrids have been reported from Olive/Anubis Baboon x Gelada Baboon (Theropithecus gelada) in the wild.
The Yellow Baboon is divdied into two races that can interbreed: P. cynocephalus cynocephalus, and P. c. kindae. The Yellow Baboon has been observed to hybridise with the Hamadryas Baboon (in captivity at Madrid and Berlin), with the Guinea Baboon (in captivity) and possibly with the Chacma Baboon (P. ursinus) (in the wild).
Hamadyas Baboon x Guinea Baboon hybrids were common in captivity, but their ranges do not overlap in the wild. The Hamadryas Baboon has also interbred with the Gelada Baboon (Theropithecus gelada ) in captivity and with the Olive/Anubis Baboon in the wild.
In captivity, the Guinea Baboon has produced hybrids with the Chacma Baboon and with the Gelada Baboon.
The Rheboon is a captive-bred Rhesus Macaque/Hamadryas Baboon hybrid with a baboon-like body shape and Macaque-like tail.
CAPUCHINS
Capuchins are widely kept in captivity and bred as exotic pets and laboratory animals (and until recently as service animals in the USA). Because they have been bred for numbers, rather than purity, for these uses many captive capuchins are hybrids or mongrels.
The White-fronted Capuchin (Cebus albifrons) has produced hybrids with the Brown-tufted (or Black-capped) Capuchin (Sapajus apella, formerly C. apella) (captivity), the Colombian White-faced Capuchin (C. capucinus) (captivity) and with the Weeper (or Wedge-capped) Capuchin (Cebus olivaceus) (captivity). The Brown-tufted (or Black-capped) Capuchin (Sapajus apella, formerly C. apella) has bred in captivity with both the Colombian White-faced Capuchin (C. capucinus) and the Weeper (or Wedge-capped) Capuchin.
The Black-striped Capuchin (C. libininosus [Black-striped) has a hybrid zone with the Black capuchin (Sapajus nigritus, formerly C. nigritus).
GIBBONS
In the primates, many Gibbons are hard to visually identify and are identified by their song. This has led to hybrids in zoos where the Gibbons were mis-identified. For example, some collections could not distinguish between Javan Gibbons, Lar Gibbons or Hoolocks and their supposedly pure breeding pairs were mixed pairs or hybrids from previous mixed pairs. Agile gibbons have also interbred with these. The offspring were sent to other Gibbon breeders and led to further hybridization in captive Gibbons. Hybrids also occur in wild Gibbons where the ranges overlap.
Because of the difficulty in identifying the different species, gibbons of different species were often accidentally paired in Western zoos. Some also hybridise in the wild and the hybrids may be almost indistinguishable from one or other parent species rather than bein intermediate in type. The following is a summary of reported hybrids.
Agile Gibbon (Hylobates agilis) x either Western Hoolock Gibbon (H. hoolock) or Pileated Gibbon (H. pileatus). The male’s identification is unclear – it most closely resembled the Western Hoolock Gibbon, but it has also been identified as a Pileated Gibbon (showing how difficult identification was before DNA testing). The hybrid offspring resembled a dark-coloured Mueller’s Grey Gibbon.
Agile Gibbon x White-handed Gibbon (H. lar) formerly had a hybrid zone in Malaysia, but this has been disrupted by logging and by an artificial lake. Agile Gibbon x Mueller’s Grey Gibbon - hybrids bred in captivity. Agile Gibbon x. Pileated Gibbon have been bred in captivity. Nine such hybrids were bred at Zurich Zoo. White-bearded Bornean Gibbon (H. albibarbus) x Mueller’s Grey Gibbon – there is a sparsely populated hybrid zone.
Black Crested Gibbon (H. concolor) x Northern White-cheeked Gibbon (H. leucogenys) – naturally occurring male and immature hybrids are almost indistinguishable from Northern White-cheekd Gibbons. Black Crested Gibbon x Pileated Gibbon – unconfirmed hybrids have occurred.
Buff-cheeked Gibbon (H. gabriellae) x Northern White-cheeked Gibbon may have hybridised (as Nomascus gabriellae x Nomascus leucogenys) giving rise to the Southern White-cheeked Gibbon (H. Soki, formerly Nomascus siki). Buff-cheeked Gibbon x Southern White-cheeked Gibbon will hybridise in the wild. Hainan Gibbon (H. hainanus) has hybridised with the Northern White-cheeked Gibbon in captivity at Twycross Zoo, producing a male hybrid offspring.
Western Hoolock Gibbon x White-handed Gibbon (H. lar) hybridise in captivity. Western Hoolock Gibbon x Eastern Hoolock Gibbon (H. leuconedys) form a cline (a type of hybrid zone)
White-handed Gibbon x Silvery Gibbon (H. moloch) hybridise in captivity. White-handed Gibbon x Mueller’s Bornean Gibbon hybridise in captivity and two adult hybrids formed a mated pair (but presumably no offspring), the female’s song combining elements from the parent species, the male’s song resembling that of the White-handed Gibbon. White-handed Gibbons will hybridise with Pileated Gibbons and with Mueller’s Grey Gibbon.
Three subspecies, Mueller's Grey Gibbon (H. muelleri muelleri), Abbott's Grey Gibbon (H. muelleri abbotti) and the Northern Grey Gibbon (H. muelleri funereus) all form hybrid zones.
Mueller's Grey Gibbon × Siamang (H. syndactylus) hybridise in captivity to form “gibbangs” or “siabons”. A female Siamang produced hybrid female "Siabon" offspring on 2 occasions when housed with a male Gibbon; one hybrid survived, the other didn't. The surviving hybrid displayed a mixture of traits from each parent: the black coat and head-hair pattern of the siamang, arm-hair pattern of the Mueller's Grey Gibbon, the large eyes of a Gibbon and long, thin nose of a Siamang. She also had Siamang-like digit webbing, but lacked the Siamang’s prominent throat sac. She had the Siamang’s deep chest, wide shoulders and wide hips, but the slender wrists, hands, ankles and feet of the Gibbon. Her limbs were proportionately shorter, as in the Siamang, but had the Gibbon’s arm-to-leg length ratio.
GUENONS
Sykes’ Monkey (Samango) has a number of races. The white-collared Cercopithecus albogularis kolbi, the white-throated C. a. monoides and an intermediate form C. a. kibonotensis that occurs between the two subspecies. Sykes’ Monkey (Cercopithecus albogularis) x Blue Monkey have a hybrid zone where the species meet, they readily hybridise in captivity. Sykes’ Monkey also hybridises with the Mona Monkey (C. mona ) and with the Vervet Monkey (Chlorocebus pygerythrus). Vervet Monkey x Sykes' Monkey (Samango) are reported in the wild and may be increasing (Yvonne A. de Jong, Thomas M. Butynski "Three Sykes's Monkey Cercopithecus mitis × Vervet Monkey Chlorocebus pygerythrus Hybrids in Kenya," Primate Conservation, 2010(25), 43-56, (1 December 2010))
At the Department of Anthropology, University of Texas, in the 1980s, an adult male Sykes’ monkey was housed with a mixed gender group of Vervet Monkeys (Chlorocebus pygerythrus) resulting in two hybrids. The facility established a mixed species group (half Vervet and half Sykes’ monkeys) including the two hybrids and their mothers. One hybrid male initially behaved socially and behaviourally like a Vervet monkey and interacted with Vervets, but was much larger than them and his appearance and behaviour increasingly resembled that of his Sykes’ Monkey sire over time.
Red-tailed Monkey (Cercopithecus ascanius) x Silver Monkey (C. doggetti) form hybrids. Red-tailed Monkey × Blue Monkey hybridise in the wild. The females are fertile and will backcross to one or other of the parent species. Red-tailed Monkey × Crowned Monkey (C. pogonias) have hybridised in captivity. Red-tailed Monkey × Moustached Guenon (C. cephus) have a hybrid zone wherein the Ngotto Guenon (C. cephus ngottoensis) has arisen as an intermediate between the two.
Moustached Guenon (C. cephus) x White-throated Guenon/Red-bellied Monkey (C. erythrogaster) appear to have given rise to an intermediate form called the Red-eared Guenon (C. erythrotis) which freely hybridises with the White-throated Guenon. Moustached Guenon x Blue Monkey may have given rise to Cercopithecus signatus (Jentink's Guenon), named by Professor Schlegel and described in Notes from the Leyden Museum, Vol VIII based on a specimen received from Rotterdam Zoological Gardens in 1877. Moustached Guenon x Mona Monkey, Moustached Guenon x Greater Spot-nosed Monkey (C. nictitans), Moustached Guenon x Lesser Spot-nosed Guenon (Cercopithecus petaurista), Moustached Guenon x Patas monkey (Erythrocebus patas) have all been described in captivity. A hybrid has been reported between the Red-eared Guenon (C. erythrotis) x Greater Spot-nosed Monkey (C. nictitans). The Greater Spot-nosed Guenon and Lesser Spot-nosed Guenon have hybridised in captivity.
The Mona Monkey (Cercopithecus mona) has hybridised with all of the following: De Brazza’s Monkey (C. neglectus), the Greater Spot-nosed Monkey (C. nictitans, the Crowned Monkey (C. pogonias and the Grivet Monkey (C. aethiops). Dent’s Mona Monkey (C. denti) x Silver Monkey (C. doggetti) hybrids occur.
White-throated Guenon/Red-bellied Monkey (C. erythrogaster) x Mona monkey (C. mona) produced a female hybrid in captivity. White-throated Guenon/Red-bellied Monkey also hybridises in the wild with Sclater’s Guenon (C. sclateri).
The Diana Monkey (C. diana) can interbreed with the Roloway Monkey (C. roloway).
The Owl-faced Guenon (C. hamlyni) produced viable female hybrids with L’Hoest’s Monkey (C. lhoesti), and produced a male hybrid with the Blue Monkey. Owl-faced Guenon x mona Monkey is also reported.
Kolb’s Blue Monkey (C. mitis kolbi) and Stuhlmann’s Blue Monkey (C. mitis stuhlmanni) are subspecies of the Nlue Monkey and freely hybridise in their contact zone. This may have given rise to two regional species, Boutourlini's Blue Monkey ( C. mitis boutourlinii) and Pousargues' Sykes' monkey (C. albogularis albotorquatus) which are both intermediate in form between Kolb’s and Stuhlmann’s Blue Monkeys.
Blue Monkey x Vervet monkey (Chlorocebus pygerythrus) - probable hybrids, generally intermediate in type, but not identical to each other, have been reported in the wild. Two captive vred hybrids were more likely to have been Blue Monkey x Tantalus Monkey (Chlorocebus tantalus).
LANGURS, LEAF MONKEYS, SURILI & COLOBUSES
The problem with this family – the Colobinae – is that taxonomists are constantly revising the nomenclatures. Several that were considered separate species have been demoted to sub-species or races which interbreed. Taxonomy is complicated by their apparent ability to interbreed with no consideration for species or sub-species boundaries.
Intergeneric hybrids are known to occur within the Colobinae subfamily. In India, Gray langurs (Semnopithecus spp.) are known to hybridize with Nilgiri langurs (Trachypithecus johnii).
Rowe, N. (1996). The Pictorial Guide to the Living Primates. Pogonias Press. pp. 139, 143, 154, 185, 223.
The Sarawak Surili (Presbytis chrysomelas) may have hybridised with the Banded Surili (P. femoralis) in the wild, giving rise to the Tricolored Surili/ Bornean Banded Langur (P. chrysomelas cruciger, previously P. arwasca). The Sarawak Surili x Maroon Leaf Monkey/Red Leaf Monkey (P. rubicunda) have been suggested as parents of the Red Banded Langur (P. melalophos cruciger), but the Tricolored Surili/ Bornean Banded Langur is now considered to be a sub-species of the Red-Banded Langur.
The Grizzled Leaf Monkey (P. comata) may interbreed with the Javan Fuscous Langur/Banded Monkey (P. fredericae), but these are now cboth onsidered sub-species of the Javan Surili (P. comata comata) and are now called P. c. comata and P. c. fredericae respectively.
Hose's langur (Presbytis hosei) may interbreed with the Maroon Leaf Monkey/Red Leaf Monkey (P. rubicunda).
The Grey-shanked Douc Langur (Pygathrix cinerea) and Red-shanked Douc Langur (P. nemaeus) appear to interbreed in the wild. The Red-shanke Douc Langur also hybridises with the Black-shanked Douc Langur (P. nigripes). The Probscis Monkey (Nasalis larvatus) has also produced hybrid offspring with the Red-Shanked Douc Langur.
A 2013 genetic study indicated that while S. entellus, S. hypoleucos, S. priam and S. johnii are all valid taxa, there is hybridization between S. priam and S. johnii, and some some hybridization in contact zones between S. entellus and S. hypoleucos, and between S. hypoleucos and S. priam. It also suggested that S. priam and S. johnii diverged from each other fairly recently.
Ashalakshmi, N.C.; Nag, K.S Chetan; Karanth, K. Praveen (2014). "Molecules support morphology: species status of South Indian populations of the widely distributed Hanuman langur". Conserv Genet.
The Southern Plains Grey Langur (Semnopithecus dussumieri) forms hybrids with the Northern Plains Grey/Hanuman Langur (S. entellus) (hybrid zone), possibly with the Tarai Grey Langur (S. hector), with the Black-footed Grey Langur (S. hypoleucos) (hybrid zone), with the Tufted Grey Langur (S. priam) (hybrid zone), with the Nilgiri/Hooded Langur (S. johnii, formerly Trachypithecus johnii) (hybrid zone), and with the Purple-faced Langur (Trachypithecus vetulus( (single captive-bred hybrid, died soon after birth).
The Northern Plains Grey Langur (Semnopithecus entellus) produced a hybrid with the Capped Langur (Trachypithecus pileatus) at the Regent’s park Zoo (London Zoo).
The Black-footed Langur (Semnopithecus hypoleucos) has a hybrid zone with the Tufted Grey Langur (S. priam) and possibly with the Nilgiri/Hooded Langur (S. johnii, formerly Trachypithecus johnii) (based on an intermediate population). The Black-footed Langur may itself derive from a hybrid population of the Southern Plains Grey Langur x Nilgiri/Hooded Langur.
The Tufted Grey Langur (Semnopithecus priam) interbreeds with the Nilgiri/Hooded Langur, the Dusky Leaf Monkey (Trachypithecus obscurus), the Capped Langur (T. pileatus) (stillborn or miscarried hybrid offspring), and the Purple-faced Langur (T. vetulus).
The Javan Langur/Javan Lutung/Ebony Leaf Monkey (Trachypithecus auratus) interbred with the Silvery Lutung/Silvered Leaf Monkey (T. cristatus) in captivity when female Javan Langurs from Bristol Zoo were sent to mate with a male at Twycross Zoo that may, retrospective to the mating, have been a Sumatran Silvery Lutung (Trachypithecus cristatus cristatus). This meant that the Javan Langur group at Bristol Zoo were hybrids and two males were destroyed as a result.
The Silvery Lutung/Silvered Leaf Monkey (T. cristatus) can breed with the Dusky Leaf Monkey (Trachypithecus obscurus), and in captivity with Phayre’s Langur/Leaf Monkey (T. Phayrei) (Calcutta Zoo, 1890s). Phayre’s Langur/Leaf Monkey may produce hybrids with the Dusky leaf Monkey where they come into contact in the wild.
The Silvery Langur (Trachypithecus cristatus) can breed with the Proboscis Monkey (Nasalis larvatus); these are distantly related species - in different genera - that share the same habitat and both have harem-based lifestyles. A female "mystery monkey" spotted in 2017 near the Kinabatangan River in Malaysian Borneo had intermediate traits between the two parent species. In 2020, this hybrid was seen nursing an infant. Proboscis monkeys are approx one third larger than Silvery langurs with pinkish faces with elongated noses. Silvery langurs have black faces with shorter, flatter noses. The hybrid has a pronounced nose like a female Proboscis monkey, but less elongated, and her face has a gray tinge. She is most likely the offspring of a male Proboscis monkey that had taken over the harem of the smaller Silver langur male.
MACAQUES

Different Macaque species can interbreed. In "The Variation Of Animals And Plants Under Domestication" Charles Darwin wrote: "A Macacus, according to Flourens, bred in Paris; and more than one species of this genus has produced young in London, especially the Macacus rhesus, which everywhere shows a special capacity to breed under confinement. Hybrids have been produced both in Paris and London from this same genus." In addition, the Rheboon is a captive-bred Rhesus Macaque/Hamadryas Baboon hybrid with a baboon-like body shape and Macaque-like tail.
Many Macaque species are inter-fertile and produce fertile offspring.
Stump-tailed Macaque (Macaca arctoides) will form hybrids with the Assam Macaque (M. assamensis) in captivity, with the Japanese Macaque (M. fuscata )in captivity and with the Rhesus macaque (M. mulatta) in captivity. In the wild, Stump-tailed Macaques and Rhesus Macaques show genetic evidence of hybridisation, and they have hybridised in captivity. The Assam Macaque has hybridised with the Long-tailed Macaque (M. fascicularis), the Pig-tailed Macaque (M. nemestrina) and Bonnet Macaque (M. radiate).
Taiwan Macaque (M. cyclopis) hybridised extensively with the Japanese Macaque (M. fuscata). The Taiwan Macaque was introduced into Japan and interbred with the native species. 3-way hybrids of Taiwan, Japanese and Long-tailed Macaque (M. fascicularis”) have been bred in captivity in Japan.
The Long-tailed Macaque (M. fascicularis) aka Crab-Eating Macaque has bred with a number of other Macques as well as with the Drill, mandrill and Yellow Baboon. There is an unverifiable report of Long-tailed Macaque x Bonnet Macaque (M. radiata). Long-tailed Macaques breed with the Philippine Macque (M. philippinensis) but these may be sub-species rather than full species.
Long-tailed Macaque x Rhesus Macaque (M. mulatta) interbreed along a hybrid zone where males of one species have crossed into the territory of the other (females stay in their family groups). The hybrids range from intermediates to varying degrees of resemblance to one or other parental species, meaning there is backcrossing occurring. Captive-bred hybrids show that some F1 hybrids are non-viable, while others survive to maturity and are able to breed. A three-way hybrid of Long-tailed Macaque x (Rhesus Macaque x Pig-tailed macaque) has also been bred. Long-tailed Macaque x Pig-tailed Macaque (M. nemestrina) hybridise in the wild. Long-tailed Macaque x Moor Macaque (M. maura) hybrids have occurred.
A hybrid between the Long-tailed Macaque x Celebes (Sulawesi) Crested Macaque (M. nigra) died soon after birth. There is a single report of a hybrid between the Long-tailed Macaque x Toque Macaque (M. sinica). Long-tailed Macaque x Lion-tailed Macaque (M. Silenus) produced a long-tailed, maneless female hybrid. Liontail Macaques have formed hybrids with Pigtail Macaques.
A male hybrid of Long-tailed Macaque x female Drill (Mandrillus leucophaeus) lived only a few hours (Hannover Zoo). The same female Drill produced a hybrid with a Moor Macaque and with a Pig-tailed macaque. A short-tailed hybrid was bred from the Long-tailed Macaque x Mandrill (Mandrillus sphinx). There is a historic account of Long-tailed Macaque) x Yellow Baboon producing a hybrid, this appears to be the same case reported as Macaca cynomolgus (Long-tailed macaque) x Hamadryas Baboon (Cynocephalus hamadryas).
The Japanese Macaque has interbred with the introduced Rhesus Macaque with the result that some apparently pure Japanese Macaques are now mongrelised with the Rhesus macaque. The Japanese Macaque has also produced hybris with the Lion-tailed Macaque.
On the island of Sulawesi, Heck’s Macaque (M. hecki) has produced hybrids with the Pig-tailed Macaque, with the Celebes (Sulawesi) Crested Macaque, with the Pig-tailed Macaque (foetus aborted), with the Gorontalo Macaque (M. nigrescens) (hybrid zone exists), with the Booted Ape (M. ochreata) and with the Tonkean Macaque (M. tonkeana) (hybrid zone exists). The Celebes (Sulawesi) Crested Macaque (M. nigra) has a hybrid zone with the Goratalo Macaque (M. nigrescens). The Celebes (Sulawesi) Crested Macaque hybrises successfully with the Lion-tailed Macaque, and with the Tonkean Macaque. The Tonkean Macaque hybridises with the Booted Ape (M. ochreata), Moor Macaque and Heck’s Macaque and has hybrid zones with each of these.
The Moor Macaque has produced hybrids with the Pig-tailed Macaque, with the Celebes (Sulawesi) Crested Macaque, with the Tonkean Macaque (M. tonkeana) (hybrid zone exists) and in captivity with the Drill (Mandrillus leucophaeus) (hydrid died soon after birth).
Moor Macaque x Tonkean Macaque hybrids may have given rise to the Togian Macaque/Balantak Monkey (M. togeana, Cynopithecus togeanus) on the Togian Islands.The Balantak Monkey is considered a hybrid swarm, rather than a distinct species, derived from female Tonkean and male Balantak genes. Field data supported the theory that the Balantak Monkey was a valid new species, but M. togeanus was a doubtful name for the population on the mainland peninsula.
Lowe, C. (2004). Making the Monkey: How the Togean Macaque Went from "New Form" to "Endemic Species" in Indonesians' Conservation Biology. Cultural Anthropology, 19(4), 491-516.
Rhesus Macaque x Pig-tailed Macaque are reported to produce fertile hybrids. Rhesus Macaque x Bonnet Macaque produced a healthy male hybrid. A female hybrid was backcrossed to a Rhesus Macaque. The Rhesus Macaque has also been crossed with the Yellow Baboon (Papio cynocephalus) and with a Hamadryas Baboon (Papio hamadryas), although only one Rhesus/Hamadryas hybrid survived out of 26 pregnancies.
The Pig-tailed Macaque is nowadays divided into two almost identical sister species, the Northern Pig-tailed Macaque (M. leonina) and the Sunda Pig-tailed Macaque (M. nemestrina) which can interbreed. The Pig-tailed Macaque (species not noted) has produced offspeing with the Celebes (Sulawesi) Crested Macaque (most hybrids non-viable, only a few reached maturity), with the Lion-tailed Macaque (M. Silenus), with the Tonkean Macaque, with the Drill (Mandrillus leucophaeus) and with the Chacma Baboon (Papio ursinus) (non-viable male hybrid).
The Bonnet Macaque has beed crossed with the Toque macaque.
The Drill (Mandrillus leucophaeus) and Mandrill (Mandrillus sphinx) have previously hybrised extensively in zoos in the past, producing fertile female hybrids, and some zoo populations contain animals with some mixed blood. The Mandrill (Mandrillus sphinx) produced a viable male hybrid with the Olive Baboon (Papio anubis), viable male and female hybrids with the Hamadryas Baboon, and also produced a female hybrid with the Yellow Baboon, but the hybrid died soon after birth. Drill × Hamadryas Baboon, and Drill x Guinea Baboon (Papio paio) have been bred, the Drill/Guinea Baboon hybrid died soon after birth.
MANGABEYS
The Lophocebus Mangabeys are closely related to Baboons. The Cercocebus Mangabeys are more closely related to the Mandrill.
The Agile Mangabey (C. agilis) has hybridised with the Grey-cheeked Mangabey (Lophocebus albigena), and with the Sooty Mangabey (C. atys) (in captivity).
The Sooty Mangabey (C. atys) will hybridise with the White-crowned Mangabey (Cercocebus lunulatus) in the wild, but F1 males and many F1 females are stillborn and there is a high incidence of stillbirth and birth defects in backcross generations. Sooty Mangabey × Red-capped Mangabey/White-collared Mangabey (C. torquatus) – male Sooty Mangabeys x female Red-capped Mangabeys in captivity have produced hybrids that rarely survive, although one lived to 20 years old. A hybrid of an albino Sooty Mangabey x female Red-capped Mangabey in 1932 had blue eyes. Male Red-capped x female Sooty reportedly died at a few days old.
The White-naped Mangabey (C. lunulatus) is now considered a subspecies of Sooty Mangabey and is intermediate in shape between the Sooty Mangabey and the Red-capped Mangabey and occupies a range between the two species. It may have hybrid origins. The White-naped Mangabey will breed with the Red-capped Mangabey In captivity. A White-naped Mangabey x Blue Monkey (C. mitis) produced two hybrids that died soon after birth.
A male Sooty Mangabey × female Long-tailed Macaque (Macaca fascicularis) produced a hybrid that survived 3 months. A male Long-tailed Macaque x female Sooty Mangabey was conceived, but the mother died in an accident during pregnancy. The same male Long-tailed Macaque mated with a mandrill (Mandrillus sphinx) which produced a viable hybrid. The Proceedings of Philadelphia Academy of Natural Sciences (1890, p. 446) lists a museum specimen that is a hybrid between Macacus cynomolgus (Long-tailed Macaque – now Macaca fascicularis) and Cercocebus fuliginosus (Sooty Mangabey – now C. atys). Sooty Mangabey × Pig-tailed Macaque (Macaca nemestrina) produced a hybrid that survived almost 2 years. Sooty Mangabey × Mandrill (Mandrillus sphinx) at Brookfield Zoo, Chicago, produced a short-tailed hybrid. A female Mangabey (Cercocebus, unspecified species) produced a male hybrid with a male Barbary Macaque (Macaca sylvanus).
A male Golden-bellied Mangabey (C. chrysogaster) produced a hybrid with an Olive Baboon (Papio anubis) at a rescue centre in 2003. This sort of liaison is not uncommon where animals are rescued as infants and raised together for mutual companionship.
Tana River Mangabey (C. galeritus) x Grey-cheeked Mangabey (Lophocebus albigena) occurred in captivity.
The Red-capped Mangabey has formed hybrids with the following species: Blue Monkey (hybrids did not survive), Long-tailed Macaque, Rhesus Macaque (Macaca mulatta), Pig-tailed Macaque (Macaca nemestrina ), Drill (Mandrillus leucophaeus) and Mandrill (Mandrillus leucophaeus). The Red-capped Mangabey x Mandrill hybrid had the Mandrill’s general colouration, but a black head, a black dorsal stripe and a long black tail. It died, aged four months, of illness.
Hybridisation of male Mangabeys (Lophocebus sp.) and female Yellow Baboons (Papio cynocephalus) resulted in hybrids called the Kipungi/Highland Mangabey. Originally assigned L. kipunji, it was later elevated to a new genus as Rungwecebus kipunji. “The kipunji is descended from a yellow baboon, and has converged on a mangabey-like morphology, or, much more likely, that it originated by hybridization between Papio cf. cynocephalus females and Lophocebus sp. males, about 0.65 Million years ago.“ Burrell, A.S., et al, “Mitochondrial evidence for the hybrid origin of the kipunji, Rungwecebus kipunji (Primates: Papionini)” Molecular Phylogenetics and Evolution, Volume 51, Issue 2, May 2009, Pages 340-348
Grey-cheeked (or White-Cheeked) Mangabey (Lophocebus albigena) and Osman’s Mangabey (L. osmani) have a hybrid zone. These were previously considered subspecies, but are now considered full species. Grey-cheeked (or White-Cheeked) Mangabey (Lophocebus albigena) x Black-crested Mangabey (L. aterrimus) produce hybrids that resembly the Black Mangabey, but with the bushy tail of the Grey-Cheeked Mangabey. Black-crested Mangabey will hybridise with Opdenbosch’s Mangabey (L. obdenboschi)
MARMOSETS
Brizilian Marmoset species are inter-fertile and form fertile hybrids. There are hybrid zones in the wild, and hybrids have been bred in captivity for the pet market where pet breeders mix the species indiscriminately. Because the hybrids appear to be fertile, this means many of the pet marmosets are probably mongrels where different species have been housed together. They are now split into 4 subgenera: Callithrix - Atlantic marmosets; Mico - Amazonian marmosets; Callibella - Roosmalens' Dwarf Marmoset; Cebuella - Pygmy Marmoset.
The Silvery Marmosets (Callithrix argentata) can breed with Common (or White-tufted) Marmosets (C. jacchus) and with the Santarem Marmoset (C. [Mico] humeralifera). Hershkovitz’s Marmoset (Callithrix (Mico) intermedia) may be derived from Silvery x Santarem hybrids.
The Buffy-tufted Marmoset (C. aurita) has a hybrid zone with the Buffy-headed Marmoset (C. flaviceps). The Buffy-headed Marmoset also has a hybrid zone with the White-headed/Geoffroy’s Marmoset, (C. geoffroyi). The White-headed/Geoffroy’s Marmoset has produced hybrids with the Common (or White-tufted) Marmoset in captivity, and one of the hybrid male offspring later sired 3-way hybrids on a Black-tufted Marmoset (C. pencillata). The White-headed/Geoffroy’s Marmoset has a hybrid zone with the Black-tufted Marmoset and their hybrids may have resulted in Wied's Marmoset (C. kuhlii) some of which are identical to captive-bred hybrids between the White-headed/Geoffroy’s Marmoset x Black-tufted Marmoset. The captive-bred hybrids have interbred with the Common Marmoset. Wied’s Black-tufted Marmoset has a hybrid zone with the Black-tufted Marmoset.
Common (or White-tufted) Marmosets (C. jacchus) have a hybrid zone with the Black-tufted Marmoset and these hybrids are common in captivity because these are the two species most commonly bred for the pet trade. Common (or White-tufted) Marmosets have also produced hybris with the Pygmy Marmoset (Cebuella pygmaea).
NIGHT MONKEYS / DOUROUCOULIS
Azara’s Night Monkey (Aotus azarae) freely interbreeds with the Feline (or Kuhl’s) Night Monkey/Owl Monkey (A. infulatus), but these have now been demoted to subspecies: Azara’s Night Monkey (A. azarae azarae), the Bolivian Night Monkey (A. a. boliviensis) and Feline Night Monkey/Owl Monkey (A. a. infulatus). This shows that the concept of species/sub-species is often a human one and not in line with nature.
ORANG-UTANS
Sumatran Orangutan (Pongo abelii) x Bornean Orangutan (P. pygmaeus) hybrids were once common in captivity because they were considered a single species. . Several hybrids were stillborn or died soon after birth. A hybid female was backcrossed to the Bornean Orangutan resulting in a stillborn male.
Sumatran Orangutan × Tapanuli Orangutan (P. tapanuliensis, identified as species in 2017) – genetic evidence shows that these had interbred in the ancient past, before becoming completely isolated from each other.
SQUIRREL MONKEYS
The Bolivian squirrel monkey( Saimiri boliviensis boliviensis) and Peruvian squirrel monkey (S. b. peruviensis) interbreed and were formely considered full species, but are now demoted to sub-species. A number of Squirrel Monkeys have overlapping ranges and hybrid-looking individuals have been observed.
The Central American Squirrel Monkey (S. oerstedii) may breed with female Common Squirrel Monkeys (S. sciureus). The Bolivian Squirrel Monkey (Saimiri boliviensis) x Common Squirrel Monkey (Saimiri sciureus) hybrids occur commonly in captivity and also in a hybrid zone in the wild. The Bolivian Squirrel Monkey also hybridises with the Black Squirrel Monkey (S. vanzolinii), these backcross with the adjacent parent species. The Common Squirrel Monkey (S. sciureus) appears to form hybrids with the Bare-eared squirrel Monkey (S. ustus) based on intermediates seen in the wild. The Bare-eared squirrel Monkey also appears to form hybrids with the Black Squirrel Monkey (S. vanzolinii), again based on the visual appearance wild-caught individuals.
TAMARINS AND LION TAMARINS
Historically, Tamarins have been divided into species based on their facial features, but this classification is now being challenged by genetic study.
A Golden-headed Lion Tamarin (Leontopithecus chrysomelas) has produced hybrids with the Golden Lion Tamarin (L. rosalia) in captivity, but the healthy pffspring died of maternal neglect.
The Pied Tamarin (Saguinus bicolor) may produce hybrids with Martins's Bare-face Tamarin (S. martinsi martinsi) in a contact zone, resulting in the Ochraceus Bare-face Tamarin (S. martinsi ochraceus). The Pied Tamarin produces fertile/partially fertile hybrids of both sexes with the Red-handed Tamarin, (S. midas).
The Brown-mantled Tamarin/Saddle-back Tamarin (S fuscicollis) has a number of sub-species that can interbreed: Red-mantled Saddle-back Tamarin (S. f. lagonotus) x Illiger's Saddle-back Tamarin (S. f. illigeri), Red-mantle Saddle-back Tamarin (S. f. lagonotus) x Andean Saddle-back Tamarin (S. f. leucogenys). The Red-mantled Saddle-back Tamarin (S. f. lagonotus) also forms hybrids with the Black-Mantled Tamarin (S. nigricollis) and with the White-mantled Tamarin (S. melanoleucus) (fertile hybrids in contact zone). Illiger’s Tamarin (S. illigeri) may also hybridise in the wild with the Black-Mantled Tamarin (S. nigricollis).
Geoffroy's Tamarin (S. geoffroyi) forms hybrids in a contact zone with the Cotton-top (Pinche) Tamarin (S. oedipus). The Red-handed Tamarin, (S. midas) has produced hybrids with the Cotton-top (Pinche) Tamarin (S. oedipus) in captivity. The White-lipped (or Red-Bellied) Tamarin (S. labiatus) has a contact zone with the Moustached Tamarin (S. mystax) where hybrids may occur.
The Emperor Tamarin (S. imperator) may have hybridised in the contact zone with the White-lipped (or Red-Bellied) Tamarin (S. labiatus). The Emperor Tamarin can form hybrids with the Moustached Tamarin (S. mystax), but the mother died from a fall before giving birth so the viability of the hybrids is unknown. The Black-chinned Emperor Tamarin (S. imperator imperator) and Bearded Emperor Tamarin (S. i. subgrisecens) interbreed in captivity, and possibly in the wild.
VERVET MONKEYS
The Grivet Monkey (Chlorocebus aethiops) has hybridised with a number of other species: historically with the Bale Mountains Vervet (C. djamdjamensis) in the wild, with the Vervet Monkey (C. pygerythrus) in a contact zone between the two species, with the Green Monkey (C. sabaeus) in captivity, with the Patas monkey (Erythrocebus patas) in captivity, with the Rhesus Macaque (Macaca mulatta) in captivity, with the Bonnet Macaque (Macaca radiate) and with the Toque Macaque (Macaca sinica). Allen’s Swamp Monkey (Allenopithecus nigroviridis) x Grivet Monkey produced a viable male hybrid in Italy.
The Vervet Monkey (Chlorocebus pygerythrus, syn. C. lalandei) has hybridised with the Green Monkey (C. sabaeus) in captivity, with the Tantalus Monkeu (C. tantalus) in the wild, with the Patas Monkey (Erythrocebus patas) in the wild and in captivity, with the Toque Macaque (Macaca sinica), with the Bonnet Macaque (Macaca radiate), and possibly with the Long-tailed Macaque (Macaca fascicularis),
The Green Monkey/Green Guenon (C. sabaeus) forms hybrids with the Tantalus Monkey, with the Patas Monkey in the wild, and with the Rhesus Macaque (Macaca mulatta) in captivity. There is a report of a male Cercopithecus callitrichus (i.e. Green Monkey) x female Macacus erythraeus (i.e. Rhesus Macaque) at the Regent’s Park Zoo in the 1870s.
Many of the Chlorocebus and Cercopithecus species interbreed at contact zones.
VARIOUS
An unknown ape (the Koolakamba) has been reported in Africa and claimed to be a Gorilla/Chimp hybrid. Larger, flatter faced, larger skulled and more bipedal than a chimp, it may also be a mutation, in which case we are witnessing evolution in action. According to von Koppenfels in 1881: “I believe it is proved that there are crosses between the male Troglodytes gorilla and the female Troglodytes niger, but for reasons easily understood, there are none in the opposite direction. I have in my possession positive proof of this. This settles all the questions about the gorilla, chimpanzee, Kooloo Kamba, N’schigo, M’bouve, the Sokos, Baboos, etc”. Yerkes reported several "unclassifiable apes" with features intermediate between chimpanzee and gorilla in his 1929 book "A Study of Anthropoid Life". In fact most of these are regional races of chimpanzee classified as separate species by over-enthusiastic naturalists.

Above: images of the koolo-kamba, n'schiego mbouve and soko from Cassell's Natural History (1901). These are now considered regional races of chimpanzee.
Garner (1896) wrote that an ape called Mafuca exhibited at Dresden Zoo in 1875 was sometimes described as a cross between chimpanzee and gorilla. Different experts identified her as a chimpanzee or as a young gorilla."It would be difficult to believe that two apes of different species in a wild state would cross, but to believe that two that belonged to different genera would do so is even more illogical. Yerkes (1929) reported the case of adult female Johanna at Lisbon, whom Duckworth (1899) considered an unclassifiable ape intermediate between gorilla and chimpanzee and similar to the “Kulu-Kamba” and Mafuca. Others considered Johanna, who had been a performing ape with Barnum and Bailey's Circus, to be a gorilla.
Here are two brief reports – press releases really – of a supposed chimp-gorilla hybrid which I suspect was really a large chimpanzee or a small gorilla. At the time there was fierce competition between animal shows to display ever more fabulous beasts to outdo their competitors. The New York Times, Saturday, May 19th, 1906 proclaimed: “BEHOLD THE GORAMPANZEE. It's a New-Fangled Hybrid Beast of Bostock'e Invention. Bostock’s Coney Island animal show is ready for business again. Bears, lions, tigers — every sort of wild beast that ever was heard of — are there in frightening numbers. A gorampanzee, even, is on band. A gorampanzee is half gorilla, half chimpanzee. It is a terrible and interesting beast.” The Daily News Democrat of Saturday March 23rd, 1907 also briefly mentioned the supposed hybrid ape: “Meantime [Bostock] has a successor in a goripanzee - hybrid producing the crossing a gorilla and a chimpanzee, a result never before obtained. Many other animals of hybrid origin will be shown.”
GORIMPHANZEE AT BOSTOCK’S. The Brooklyn Citizen, 18th May 1906, pg 4.
Is a Cross Between Gorilla and Chimpanzee. The most remarkable animal ever seen and the only one of its kind is the Gorimphanzee which will b shown at a private view at Bostock’s wild animal show at Coney Island to-night. Mr. Bostock has issued a limited number of invitation for the event. The Gorimphanzee, so named by Bostock, is a cross between a chimpanzee and a gorilla, and is the only hybrid of its kind in existence. The result was obtained by investigations conducted by a well-known Parisian scientist who experimented with several apes in the hope of obtaining some clew to the ordinary theory of evolution. The animal is a female and is mature. She has the face of a chimpanzee, but the general characteristics of the gorilla. She and a single chimpanzee are the only survivors of Bostock’s remarkable family of anthropoid apes, which he hoped to exhibit this summer. The others died at sea on the trip over.
FRANK BOSTOCK’S EXHIBIT. The Brooklyn Daily Eagle (New York). 19th May 1906, pg 12.
The center piece of attention last night was the Gorimphanzee, the hybrid gorilla-chimpanzee, which is the only one known to be in existence. Several well-known physicians were present by invitation to view this freak. It was shown together with a chimpanzee, and is about twice the size of the latter.
According to the Deadwood Pioneer Times (4th October, 1934) a “hybrid ape, half gorilla and half chimpanzee, has been reported seen several times in Africa, says Dr. William M. Mann, director of the National zoological park. African natives persistently tell of the ‘Koddoelo,’ a baboon-like creature big as a man. Sometimes it walks on two legs, sometimes on four. When it approaches, natives desert their villages and cross the nearest river for safety.”
CHIMP / BONOBO HYBRIDS
The Congo and the Lualaba rivers form a physical barrier between bonobos and common chimpanzees. Bonobos and common chimps are genetically and behaviourally different enough to be considered separate species. There are no accounts of hybridisation under natural conditions, however genetic studies show that bonobos have donated genes to chimps more than once in the 1.5 million to 2.1 million years since they last shared a common ancestor.
In captivity, when accidentally mixed with chimps, bonobos - being less aggressive - end up at the bottom of the pecking order. Common chimps form same-sex coalitions, while bonobos seem far less political, using recreational sex to cement bonds within the troupe. Usually the dominant chimp male mates with a selection of high-ranking females. The less aggressive bonobo males do not become high-ranking enough to challenge the alpha for mating rights or to attract the attention of females. Female bonobos would also be low-ranking and unlikely to attract the attention of the alpha male. The bonobos also retain the tail tuft that chimps interpret as a juvenile trait. Mating between the two species would occur when a male of one species is housed with females of the other species.
Bonobos and chimps have mated in captivity (Savage-Rumbaugh, S. and B.J. Wilkerson, 1978. “Sociosexual behavior in Pan paniscus and Pan troglodytes: a comparative study.” Journal of Human Evolution, 7: 327-344)
Some hybrids between the two species have described (H. Vervaecke H. & Van Els Acker, L. “Hybrids between common chimpanzees (Pan troglodytes) and pygmy chimpanzees (Pan paniscus) in captivity” Mammalia, January 1992.) These hybrids and their parents lived with the Rech family.
Three hybrids were sired by a bonobo male, Congo, and two common chimpanzee females, Julie and Clara. All 3 parents were probably born between 1978 and 1980; the females had no other offspring except the hybrids. Congo had been housed with a common chimp female, Zora, of his own age since they were both one year old. Congo and Zora never mated. Julie and Clara were housed in an adjacent cage and Congo mated with them whenever he had access to them. Five pregnancies resulted.
In 1990, Congo x Clara resulted in an aborted foetus at 6 months gestation.
In 1990, Congo x Julie resulted in an aborted 2 – 3 month embryo.
25-March-1991 – Congo x Julie produced male hybrid, Blois.
5-December-1991 – Congo x Clara produced female hybrid, Tibie.
10-Martch-1992 – Congo x Clara produced female hybrid, Diana.
Compared to common chimpanzees, the hybrid infants had smaller, darker ears, a darker and less protruding lower jaw, pink lips and marked whiskers (baby common chimps have pale faces, while baby bonobos often have dark faces). The female genitalia were larger and more ventrally (towards the front) oriented. Compared to a bonobo male, the one year old male hybrid had a wider chest and spotted pigmentation on the inner side of the thighs. None of the hybrids had a webbed second and third toe. The hybrids were hand-raised by the Rech family and had restricted auditory contact with the adults. Their vocalisations were analysed and had some characteristics of both species. Compared to common chimps hand-reared by the family, the one-year-old male hybrid was less aggressive, did not avoid water and often (and easily) walked bipedally. The hybrids continued to be studied.
(H. Vervaecke H. & Van Els Acker, L. “Hybrids between common chimpanzees (Pan troglodytes) and pygmy chimpanzees (Pan paniscus) in captivity” Mammalia, January 1992)
The hybrids continued to thrive and updated information described four bonobo-chimpanzee hybrids (2 males, aged 10 and 9, and 2 females aged 10 and 8). As adults, they varied in the degree of expression of chimpanzee or bonobo features e.g. the slender/sturdy conformation, degree of protruding lower jaw, dorsal/ventral position of female genitalia. There seemed to be more female-oriented interactions, compared to the male-dominated, aggressive behaviour found in the common chimp. Vocalisations included the chimpanzee pant-hoot, but higher-pitched and with bonobo inflections.
HUMANZEES (& OTHER CLAIMED HUMAN HYBRIDS)
A reputed "humanzee" (human/chimp hybrid) called Oliver was DNA tested and found to be a chimpanzee, albeit one which slightly differed genetically from the more familiar chimps in being bipedal and having a smaller head. Oliver may have been a mutant or represent an unknown species of ape. It is currently believed that he represents a geographical subspecies of chimpanzee. He did not associate with other chimps in captivity as was sexually attracted to human women instead. This meant he was never bred. Oliver's habitual bipedal gait is now believed to be a result of early training and habit, although he mastered it to a greater degree than most trained chimps. It's worth remembering that evolution is a never-ending process and that it's possible for bipedalism to develop in other apes. In a publicity event, a woman declared her willingness to be inseminated by Oliver (and even to have the mating filmed for scientific purposes), but this offended public sensibilities and did not happen. Had Oliver been a genuine hybrid, then like most male hybrids he would probably have been sterile anyway.
Soviet Professor Ilya Ivanov attempted to create a human-ape hybrid using female chimps impregnated and human sperm and planned to use women volunteers impregnated with chimp sperm. Ivanov's experiments have been documented by Kirill Rossiianov (Institute for the History of Science and Technology of the Academy of Sciences, Moscow), "Beyond Species: Ilya Ivanov and His Experiments on Cross-Breeding Humans with Anthropoid Apes," Science in Context, 2002, p. 277-316.
In a presentation to the World Congress of Zoologists in Graz in 1910, he outlined the possibility of using artificial insemination to create a hybrid. In 1924, while working at the Pasteur Institute in Paris, Ivanov gained permission from the Institute's directors to use its experimental primate station in Kindia, French Guinea, for his hybridisation experiments. He requested backing for this project from the Soviet government, writing to Soviet officials including the People's Commissar on Education and Science Anatoliy Vasilievich Lunacharsky. In September 1925, Nikolai Petrovich Gorbunov, head of the Department of Scientific Institutions helped allocate US$10000 to the Academy of Sciences for Ivanov's human-ape hybridization experiments in Africa.
In February 1926, Ivanov set off for Africa, via Paris, where the directors of the Pasteur Institute seemed enthusiastic and promised him access to the chimps at the Institute’s new primate centre in French West Africa (now Guinea). In March 1926 Ivanov arrived at the Kindia facility, but left after a month because the facility had no sexually mature chimpanzees. Ivanov attempted to organize the insemination of human females with chimpanzee sperm in Guinea, but the French colonial government objected to the proposal. There is no evidence such an experiment was arranged there. Ivanov planned to return later that year to capture his own chimps.
Back in Paris, he spent time at the Pasteur Institute and worked with surgeon Serge Voronoff. Voronoff grafted slices of ape testes into the testes of older men who wanted to be “rejunvenated.” Ivanov also transplanted a woman’s ovary into a chimp called Nora and then inseminating Nora with human sperm. While in France he corresponded with French Guinea's colonial governor and arranged to conduct his experiments at the botanical gardens in Conakry. Ivanov, assisted by his son (also called Ilya), went to Conakry in November 1926 where he oversaw the capture of adult chimpanzees in the interior of the colony. These were caged at the botanical gardens in Conakry. On February 28, 1927, Ivanov artificially inseminated 2 female chimps with human sperm (not sourced from him or his son), apparently with difficulty. On June 25, he injected a third chimpanzee with human sperm. He also planned a second experiment: to inseminate women with chimp sperm, but aware that no local woman would agree to this, he planned to do so under the pretext of a medical examination. The French governor forbade this. The Ivanovs left Africa in July 1927 with 13 chimps, including the 3 artificially inseminated females. They already knew that the first 2 chimps had not conceived. The third died in France and was also found not to have conceived. The remaining 10 chimps went to the Sukhumi primate station.
Ivanov returned to the Soviet Union in 1927 and attempted to organize experiments at Sukhumi using ape sperm and human females. He took 20 chimps to create an ape nursery in Abkhazia, only 4 of which actually made it, and realised that his best chance of creating a hybrid was to recruit Soviet women willing to be inseminated with chimp sperm.
In 1929, with the help of Gorbunov, he gained the support of the Society of Materialist Biologists (a group associated with the Communist Academy). In Spring 1929 the Society set up a commission to plan Ivanov's experiments at Sukhumi. They required at least 5 volunteer women for the project. In June 1929, before any inseminations had taken place, the only sexually mature ape remaining at Sukhumi (a 26 year old orangutan called Tarzan) had died. A new set of chimps would not arrive at Sukhumi until Summer 1930. That year, a political shakeup in the Soviet scientific world resulted in Gorbunov and several other Sukhumi scientists losing their positions. In Spring 1930 Ivanov came under political criticism and on December 13, 1930 he was arrested and exiled to Alma Ata, where he died in 1932.
One wonders why Ivanov wanted to produce ape-human hybrids and why it was initially encouraged. If successful, as well a being a Soviet scientific coup, it would support Darwin’s theory of evolution and help the Bolsheviks stamp out religion. It would also prove that artificial insemination – using the sperm of selected men - could be used to improve humans for the benefit of the socialist state.
There have been persistent rumours of a Chinese humanzee experiment; the rumoured 3 month foetus died when the mother was killed during civil unrest. Allegedly scientists impregnated a female chimpanzee with human sperm and she was three months pregnant before people found out; outraged they broke into the lab in a riot, killing the pregnant chimpanzee. This must be treated as urban myth as there is no currently no evidence to support the tale and far to many claims of conspiracy theory cover-ups.
There are similar rumours of a humanzee or manpanzee experiment in the USA. In the 1960s there were persistent rumours of a Russian experiment to inseminate either a female chimpanzee or a female gorilla with human sperm. Bernard Grizmek, former Frankfurt Zoo director, wrote of rumours from the Soviet Union that the Russians had created a human/chimpanzee hybrid (probably a mis-reporting of Ilya Ivanov's experiments). More recently, a news story claimed that Stalin ordered his scientists to create an army of human/ape hybrids, because they would be less fussy about what they ate. Though nothing came of this, it may have been the origin of the rumours.
According to a tale by Peter Damain in the 11th century story “De bono religiosi status et variorum animatium tropolagia,” Count Gulielmus had both a pet ape and a wanton wife. The woman was so wanton that she allowed the ape to become her lover. The ape became jealous of the Count and when it found him lying with the Countess, the ape attacked him. The Count died of his grievous injuries. Damain had learned of this from Pope Alexander II. The pope had shown Damain a monster that was supposedly the result of the ape mating with the woman. This apelike boy was called Maimo after his simian father. If Maimo did exist, he was most likely a physically and mentally handicapped child.
In the 19th century, a Khoisan (Hottentot) woman called Saartjie Baartman was exhibited in Europe in a cage. Negro women with enlarged labia and enlarged buttocks were sometimes deemed evidence of chimp/human hybridisation; such hybrids being called a "womanzee". This was based on the supposed resemblance of their genitalia to those of female chimps and fitted with the then prevalent opinion that Negroes were inferior, or less evolved, than Europeans. Enlarged buttocks occur due to a condition called steatopygia (extreme accumulation of fat on the buttocks), while enlarged labia, or "Hottentot Apron" can be either inherited or induced/enhanced by manual stretching (in some regions they were considered attractive). Neither trait is due to hybridisation.
The idea of human/ape hybrids has fascinated people and resulted in several films or TV series, some exploring whether such hybrids would have "human rights" or simply be experimental animals for use in vivisection. It is only a matter of time before curiosity overcomes ethics and an authenticated attempt is made.
Textual content is licensed under the GFDL.
|
BACK TO HYBRID & MUTANT BIG CATS INDEX |